Abstract
Introduction
The ACCM/PALS guidelines address early correction of paediatric septic shock using conventional measures. In the evolution of these recommendations, indirect measures of the balance between systemic oxygen delivery and demands using central venous or superior vena cava oxygen saturation (ScvO2 ≥ 70%) in a goal-directed approach have been added. However, while these additional goal-directed endpoints are based on evidence-based adult studies, the extrapolation to the paediatric patient remains unvalidated.
Objective
The purpose of this study was to compare treatment according to ACCM/PALS guidelines, performed with and without ScvO2 goal-directed therapy, on the morbidity and mortality rate of children with severe sepsis and septic shock.
Design, participants and interventions
Children and adolescents with severe sepsis or fluid-refractory septic shock were randomly assigned to ACCM/PALS with or without ScvO2 goal-directed resuscitation.
Measurements
Twenty-eight-day mortality was the primary endpoint.
Results
Of the 102 enrolled patients, 51 received ACCM/PALS with ScvO2 goal-directed therapy and 51 received ACCM/PALS without ScvO2 goal-directed therapy. ScvO2 goal-directed therapy resulted in less mortality (28-day mortality 11.8% vs. 39.2%, p = 0.002), and fewer new organ dysfunctions (p = 0.03). ScvO2 goal-directed therapy resulted in more crystalloid (28 (20–40) vs. 5 (0–20) ml/kg, p < 0.0001), blood transfusion (45.1% vs. 15.7%, p = 0.002) and inotropic (29.4% vs. 7.8%, p = 0.01) support in the first 6 h.
Conclusions
This study supports the current ACCM/PALS guidelines. Goal-directed therapy using the endpoint of a ScvO2 ≥ 70% has a significant and additive impact on the outcome of children and adolescents with septic shock.
Similar content being viewed by others
Introduction
Severe sepsis and septic shock are major causes of morbidity, mortality and resource consumption. Although outcomes have improved over the last few decades [1–4], mortality rates of children with severe sepsis reach 20% in developed nations [5–8], are higher than 50% in developing countries [8–11], and even higher among patients with underlying comorbidities and organ dysfunction [7, 12–15].
Early recognition and aggressive fluid resuscitation have a crucial role in the treatment of paediatric septic shock [3, 16]. The American College of Critical Care Medicine – Paediatric Advanced Life Support (ACCM/PALS) guidelines recommend rapid, stepwise interventions with the following therapeutic endpoints in the first hour: capillary refill of < 2 s, normal pulses with no differential between peripheral and central pulses, warm extremities, urine output > 1 ml/kg/h and normal mental status. Further haemodynamic optimisation using metabolic endpoints to treat global tissue hypoxia include a superior vena cava oxygen saturation (ScvO2) ≥ 70% and cardiac index > 3.3 and < 6.0 l/min/m2 with normal perfusion pressure for age [17]. The evidence which has resulted in these current guidelines has been derived from limited prospective and retrospective studies or case series [3, 18, 19]. More specifically, the outcome benefit of further optimising metabolic parameters, namely ScvO2, in goal-directed therapy remains unknown.
Although current ACCM/PALS guidelines represent best practice, prospective randomised trials are lacking to confirm all components of these recommendations. The purpose of this study was to examine whether treatment according to ACCM/PALS guidelines performed with and without goal-directed therapy to achieve a ScvO2 ≥ 70% reduces the morbidity and mortality rate of children with severe sepsis and septic shock [20].
Materials and methods
The ethics committees of both hospitals involved approved this prospective, randomised controlled trial, as well as the written informed consent. Interim analysis was conducted by a Safety and Monitoring Review Committee under the direction of the ethics committees.
Participants
Patients were recruited from the emergency department, the inpatient unit and the paediatric intensive care unit (PICU) of two hospitals affiliated to the University of São Paulo, Brazil, from January 2004 to August 2005. The first is a 245-bed tertiary-care paediatric hospital, with 13 beds in the PICU; the second is a 308-bed secondary-care general hospital, with 10 beds in the PICU. We included patients with severe sepsis or fluid-refractory septic shock that had not responded after 40 ml/kg of any resuscitation fluid or that required cardiovascular agents at any time during resuscitation. Severe sepsis was defined as sepsis (evidence of infection and two of the following: 1, hypothermia or hyperthermia; 2, tachycardia or bradycardia if < 1 year old; 3, tachypnoea; 4, white blood cell count > 12,000 or < 4,000 cells/ml3 or > 10% immature form) and at least one of the following indications of altered organ function or hypoperfusion: altered mental status, hypoxaemia, increased serum lactate level, bounding pulses, oliguria (< 1 ml/kg/h) or hypotension [17, 21]. The criteria for exclusion from the study were refusal to sign the written informed consent, age less than 1 month or more than 19 years, uncorrected cyanotic heart disease, exclusive palliative care, and arrival from another hospital more than 6 h after the diagnosis of severe sepsis or septic shock.
Randomisation
After written informed consent had been obtained from a next of kin or legal guardian, patients were randomly assigned to either the intervention or the control group. A computer-generated random sequence was placed in sealed, opaque envelopes, kept at the first hospital. For each included patient, in any of the two hospitals, the next envelope was opened, assigning that patient to intervention or control group.
Procedures
The patients assigned to intervention received a catheter capable of measuring ScvO2 (Edwards Lifesciences, Irvine, CA), which was connected to a monitor exclusively for continuous ScvO2 monitoring. The catheter used is 4 Fr, 40 cm long, with one infusion lumen and one optic fibre, and was inserted through a percutaneous sheath introducer (5 Fr for infants and 6 Fr for children and adolescents), that has another infusion lumen [22]. The ScvO2 monitor was calibrated after catheter placement and once daily after that. ScvO2 was continuously displayed and values were confirmed by venous blood analysis at 0,6 and 72 h. The patients assigned to control received a double-lumen central venous catheter (5 Fr for infants and children and 7 Fr for adolescents), and ScvO2 was measured by venous blood analysis (values were recorded, but not used to guide treatment). In both groups, the catheter was placed with its tip inside the right atrium or at the superior or inferior cava junction, close to the right atrium. Catheter position was confirmed by chest radiography. In both groups, central lines were placed by the fellow or attending physician responsible for the patient's care.
The medical team was usually composed of two residents, one fellow and one attending physician. The patients assigned to the control group received ACCM/PALS therapies without continuous ScvO2 monitoring, with fluid resuscitation (crystalloid or colloid), red blood cells and cardiovascular agents directed to maintain normal perfusion pressure for age, urine output > 1 ml/kg/h, capillary refill of < 2 s and normal pulses (Fig. 1).
The patients assigned to the intervention group received ACCM/PALS therapies directed toward the endpoint of ScvO2 ≥ 70% using continuous monitoring. If the ScvO2 was < 70%, even with normalisation of perfusion pressure, urine output and peripheral perfusion, then more fluid, red blood cells (if haemoglobin < 10 g/dl) or inotropes were given (Fig. 1). The treatment was considered to be successful if the patient showed normal blood pressure for age, normal pulses, warm extremities, urine output > 1 ml/kg/h and ScvO2 ≥ 70%.
In both groups, other supportive therapies, such as mechanical ventilation, nutrition, antibiotics, and renal replacement therapy, were decided by the medical team, according to the routine practice. The duration of the protocol resuscitation was 72 h in each group.
Baseline characteristics of the patients were recorded including age, gender, underlying comorbidity, site of infection, time elapsed between hospital admission and the diagnosis of severe sepsis or septic shock, and the amount of resuscitation fluid and cardiovascular agent received prior to the study entry. The patients' heart rate, respiratory rate, arterial blood pressure, arterial oximetry, central venous pressure and oxygen saturation, urine output, PRISM score (PRISM) and Multiple Organ Dysfunction Score (MODS) [23, 24] (with the laboratory tests required to calculate the score) were obtained at base line (0 h) and after 6 and 72 h. The amount of resuscitation fluid (crystalloid and colloid) and red blood cells administered, and the use of cardiovascular agents and steroids, were registered during the intervals from 0 to 6 h and from 6 to 72 h after the beginning of the treatment. Patients were followed for 60 days or until hospital discharge or death.
Statistical analysis
Twenty-eight-day mortality was the primary endpoint. Secondary endpoints were number of organ dysfunctions, administered treatments, duration of therapy with cardiovascular agents, duration of mechanical ventilation, length of PICU stay, days free of cardiovascular agents and days free of mechanical ventilation.
Categorical variables are presented as percentages. Normally distributed continuous variables are presented as means with standard deviations; and skewed data are presented as median and interquartile range (25–75%). Categorical variables were compared using Fisher's exact test. Normally distributed continuous variables were analysed with Student's t-test, and non-normally distributed variables with the Mann–Whitney rank-sum test. All tests were two-sided, and a p-value of less than 0.05 was considered to indicate a statistically significant difference. Kaplan–Meier estimates of mortality, along with hazard ratios and 95% confidence intervals, were used to describe the relative risk of death.
A multivariate logistic regression model was developed to examine the odds ratios (OR) and 95% confidence intervals of variables associated with 28-day mortality. Variables with a univariate association (p < 0.10) and variables with clinical plausibility were introduced into a backward stepwise multiple linear regression. Variables were retained in the regression model if they were significant at a level of 0.05. Renal failure (0 or 1), neurological failure (0 or 1), group (0 or 1), age, pH at baseline and PRISM score were included as co-variants.
Assuming an alpha value of 0.05 and a power of 80%, we calculated that a sample size of 268 patients was necessary to permit the detection of a 35% relative reduction or 20% absolute reduction in mortality from 57.3%, which was our baseline mortality. The protocol included interim analysis after the enrolment of 100 patients, which was independently conducted by the ethics committee.
Results
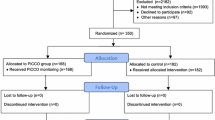
During the study period, 110 patients were eligible and 8 patients (7.3%) were excluded (seven arrived from other hospitals more than 6 h after severe sepsis diagnosis and one had uncorrected cyanotic heart disease). The 102 patients enrolled were randomly assigned to either the intervention or the control group and were included in the intention-to-treat analyses (Fig. 2). The study was terminated after the interim analysis, following the recommendation of the ethics committee of our hospital.
Baseline characteristics were similar between the two groups, with the exception that the intervention group was older (Table 1). The primary endpoint, 28-day mortality rate, was significantly lower in the intervention group (hazard ratio (HR) = 3.78 (1.58–7.52); p = 0.002), with a number needed to treat of 3.6 (Table 3). Multivariate logistic regression analysis showed that PRISM score (OR = 1.20 (1.06–1.36); p = 0.006), and renal dysfunction (OR = 7.76 (1.86–32.33); p = 0.005) were independently associated with increased 28-day mortality, whereas a goal-directed resuscitation using ScvO2 ≥ 70% as an endpoint was independently associated with decreased 28-day mortality (OR = 0.18 (0.05–0.69); p = 0.01). Age, pH at baseline and neurological dysfunction were not independently associated with 28-day mortality.
Considering all patients, 28-day mortality was higher for patients who presented with ScvO2 < 70% (41.9% vs. 24.3%; HR = 3.04 (1.43–8.79); p = 0.006). Comparing the control versus the intervention group, mortality was similar in the two groups for patients with ScvO2 ≥ 70% (21.7% vs. 11.8%; HR = 1.91 (0.51–7.57); p = 0.32), but significantly different for patients who presented with ScvO2 < 70% (68.8% vs. 13.3%; HR = 6.83 (1.80–17.02); p = 0.003) (Fig. 3).
The number of organ dysfunctions at enrolment was similar in the two groups. The intervention group developed fewer new organ dysfunctions, due to reduced incidence of renal and neurological dysfunction (Table 3). Before study entry, the initial resuscitation was similar in the two groups: the control group received 40 ml/kg (interquartile range 30–70 ml/kg) of crystalloid, no colloid, and 5.9% of the patients received packed red blood cells (PRBC), while the intervention group received 40 ml/kg (interquartile range 30–70 ml/kg) crystalloid (p = 0.89), no colloid (p = 1.00), and 3.9% received PRBC (p = 1.00). Between 0 and 6 h after study entry, the intervention group received significantly more crystalloids (p < 0.0001) and more frequently received red-cell transfusion (p = 0.002) and inotropic support (p = 0.01) (Table 4).
Other supportive therapies were similar between the two groups (Table 4). Pulmonary artery catheters and activated protein C were not used in any patient, and extracorporeal membrane oxygenation was not available in either of the two centres involved in the trial. All patients enrolled in the study received a central line for at least 72 h and there was no complication related to line placement.
Discussion
In this randomised controlled trial, there was a significant difference in mortality rate with use of ACCM/PALS haemodynamic support guidelines for septic shock between patients with or without ScvO2 guided therapy. Patients who received therapies directed to the goal of ScvO2 > 70% were given more fluid, red blood cells and inotropic support after the initial resuscitation, with a resulting 3.3-fold reduction in mortality. This study supports the current ACCM/PALS guidelines. Goal-directed therapy using the endpoint of a ScvO2 ≥ 70% provided a significant and additive impact on the outcome of children and adolescents with septic shock in our centres.
In 2002, consensus guidelines on haemodynamic support of paediatric and neonatal patients with septic shock recommended that early recognition, adequate fluid resuscitation, appropriate cardiovascular therapies, and timely and appropriate antibiotic therapy and source control are all crucial to optimal outcome [17]. Studies in children have shown that early aggressive volume repletion, even without addressing myocardial dysfunction, significantly improves outcome [16, 25, 26]. Similar findings have been shown when conventional ACCM/PALS guidelines are applied in the emergency department setting before PICU admission. More importantly, when the lack of shock reversal secondary to inadequate resuscitation was present, the odds of mortality doubled with each hour of persistent shock [25]. Recent adult studies have shown that goal-directed resuscitation using SvO2/ScvO2 has pathogenic, prognostic, therapeutic and outcome benefits in the management of severe sepsis and septic shock [27, 28]. However, prior to this study, the addition of ScvO2 as an endpoint in PALS, while making physiological sense, had not been validated in an outcome study.
Goal-directed haemodynamic optimisation represents an organised approach to volume repletion, attainment of a target blood pressure (vascular tone) and a more definitive resolution of a pathologic oxygen supply dependency or global tissue hypoxia through a normalisation of ScvO2 as a surrogate endpoint. A study in adult patients with severe sepsis and septic shock confirmed that global tissue hypoxia, defined by an ScvO2 < 70%, can still be present even if physical examination, vital signs, central venous pressure and urinary output are considered adequate. In addressing this global tissue hypoxia using an early goal-directed approach, further reductions in the inflammatory response, morbidity and mortality were achieved compared with conventional therapy [27]. While the early goal-directed therapy patients received more fluids, red blood cell transfusions and inotropes over the first 6 h for a higher rate of attaining a ScvO2 ≥ 70%; there was essentially no difference in these therapies after the first 72 h. Because of this early haemodynamic optimisation, significant reductions in vasopressors, mechanical ventilation and pulmonary artery catheter use were also realised [27].
Similar to this previous study, the mortality reduction observed in our study is most likely the result of multiple interventions. Patients who had failed initial attempts at volume resuscitation based on ACCM/PALS recommendations was equally distributed between the two treatment groups and provided a high degree of illness severity. In spite of considerable initial volume resuscitation, the intervention group received more fluid (crystalloid), more red blood cell transfusion and more inotropic support in the first 6 h of treatment than the control group. After 72 h, the amount of crystalloid administered and the percentage of red blood cells transfusion were not different between the two groups. These observations were similar to those found in adults. Together, these two studies support the concept that early resuscitation rather than later resuscitation is beneficial [17, 18, 25, 26]. In addition, there was a statistically significant reduction in the number of children with ScvO2 < 70% at 72 h in the intervention group. This supports the concept that goal-directed therapy can reduce global oxygen debt over the long term as well as the short term in patients with septic shock.
The issue of whether an ScvO2 of 70% is an adequate therapeutic endpoint for the resolution of global tissue hypoxia has been heavily debated in adult, paediatric and neonatal models [29–37]. Studies in this regard are lacking in children, which makes the recommended value of 70% as target value in ACCM/PALS guidelines empirical [17]. Furthermore, the evaluation of the technology of continuous venous oximetry is particularly important in developing countries because of the potential added cost to health care. Our group previously has examined the clinical utility of this technology in the paediatric patient [22].
ScvO2 proved to be beneficial in discriminating high-risk patients. Kaplan–Meier estimates showed a higher mortality rate for patients with ScvO2 < 70% at presentation. The similarity between the curves for ScvO2 ≥ 70% in both groups and the significant difference between the curves for ScvO2 < 70% in the two groups supported the hypothesis that the intervention rescued patients from cryptic shock, and provided validation of the true independence of the treatment protocol from assignment groups.
Although a considerable proportion of children presented with ScvO2 below 70%, the average values were not as low as those reported in adults. Since very few data derived in similar circumstances are available in the literature, we can only speculate that children with severe sepsis were diagnosed earlier than adults or that unspecified age-dependent factors explain the observation. The observed absence of increased lactate values was consistent with less dramatic reductions in ScvO2 in our children than those reported in adults.
In addition to the favourable reduction in mortality, a decrease in pulmonary (increased number of ventilator-free days), neurological and renal dysfunction was also seen. These findings support previous studies showing that early haemodynamic optimisation is associated with decreased health care resource consumption [38, 39]. Because of these salutary findings, the intervention group protocol has now become a standard of care at both participating hospitals.
Limitations
Several limitations are important to consider when evaluating this study. First, this was not a blinded study as the catheter and spectrophotometry unit used to monitor ScvO2 was not present in the control group. To minimise this influence, all treatment decisions were based on ACCM/PALS guidelines and were decided by the medical team responsible for the patient. Second, although ScvO2 monitoring may provide an early warning signal that may prompt earlier interventions before haemodynamic deterioration [28], this study is not an endorsement of continuous venous oximetry technology. In the absence of continuous monitoring, intermittent sampling may provide benefit. Third, the mortality rate observed in the control group is similar to or even lower than those reported in studies from developing countries, especially when patients with underlying diseases and multiple organ failure were included, and it was also lower than the mortality rate reported by our group two years before the study (39.2% vs. 57.3%), possibly reflecting a treatment effect of ACCM/PALS guidelines even without ScvO2 monitoring. Fourth, baseline characteristics of the two groups were similar, except for age, with the control group population being younger. Even though the multivariate logistic regression analysis showed there was no difference in outcome attributable to age, and previous studies from our centres reported that increased age was associated with increased mortality, this baseline difference should still be kept in mind [19]. Fifth, we studied a population which had a mortality rate of 40%. A recent survey of the KIDS database in the United States showed an 8% mortality rate for children with severe sepsis and chronic illness, and a 2% mortality rate for those with severe sepsis who were previously healthy [40]. Although validation of the presented data would be desirable for different populations, we believe it is crucial to have a resuscitation target in any environment, as we observed that the benefit arose from rescuing patients from cryptic shock, rather than offering a stimulus to more aggressive treatment. At this moment, the presented results are valid only for this sample of patients. Sixth, the trial was conducted in two centres in Brazil, indicating that the findings are specifically valid for our country and possibly other developing nations, where health care resources are limited and more sophisticated technologies are not widely available. It remains unknown whether this technology will be beneficial in technology-rich environments.
Conclusion
We conclude that goal-oriented resuscitation with the current ACCM/PALS guidelines can improve morbidity and mortality when supplemented by ScvO2 monitoring. These findings may have a significant impact on the outcome of children and adolescents with septic shock.
References
DuPont HL, Spink WW (1968) Infectious due to gram negative organisms: an analysis of 860 patients with bacteremia at University of Minnesota Medical Center, 1958–1966. Medicine (Baltimore) 48:307–311
Pollack MM, Fields AI, Ruttimann UE (1985) Distributions of cardiopulmonary variables in paediatric survivors and nonsurvivors of septic shock. Crit Care Med 13:454–459
Carcillo JA, Davis AL, Zaritsky A (1991) Role of early fluid resuscitation in paediatric septic shock. JAMA 266:1242–1245
Watson RS, Carcillo JA, Linde-Zwirble WT, Clermont G, Lidicker J, Angus DC (2003) The epidemiology of severe sepsis in the United States. Am J Respir Crit Care Med 167:695–703
Pollard AJ, Britto J, Nadel S, DeMunter C, Habibi P, Levin M (1999) Emergency management of meningococcal disease. Arch Dis Child 80:290–296
Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR (2001) Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med 29:1303–1310
Kutko MC, Calarco MP, Flaherty MB, Helmrich RF, Ushay HM, Pon S, Greenwald BM (2003) Mortality rates in paediatric septic shock with and without multiple organ system failure. Pediatr Crit Care Med 4:333–337
Khilnani P, Sarma D, Zimmerman J (2006) Epidemiology and peculiarities of pediatric multiple organ dysfunction syndrome in New Delhi, India. Intensive Care Med 32:1856–1862
Sarthi M, Lodha R, Vivekanandhan S, Arora NK (2007) Adrenal status in children with septic shock using low-dose stimulation test. Pediatr Crit Care Med 8:23–28
Chang P, Hsu HY, Chang MH, Lin FY (1999) Shock in the paediatric emergency service: five years' experience. Acta Paediatr Taiwan 40:9–12
Branco RG, Garcia PC, Piva JP, Casartelli CH, Seibel V, Tasker RC (2005) Glucose level and risk of mortality in paediatric septic shock. Pediatr Crit Care Med 6:470–472
Wilkinson JD, Pollack M, Glass NL, Kanter RK, Katz RW, Steinhart CM (1987) Mortality associated with multiple organ system failure and sepsis in paediatric intensive care unit. J Pediatr 111:324–328
Proulx F, Fayon M, Farrell CA, Lacroix J, Gauthier M (1996) Epidemiology of sepsis and multiple organ dysfunction syndrome in children. Chest 109:1033–1037
Plotz FB, Hulst HE, Twisk JW, Bökenkamp A, Markhorst DG, van Wijk JA (2005) Effect of acute renal failure on outcome in children with severe septic shock. Pediatr Nephrol 20:1177–1181
Pancera CF, Costa CM, Hayashi M, Lamelas RG, Camargo B (2004) Severe sepsis and septic shock in children with cancer. Rev Assoc Med Bras 50:439–443
Booy R, Habibi P, Nadel S, de Munter C, Britto J, Morrison A, Levin M, Meningococcal Research Group (2001) Reduction in case fatality rate from meningococcal disease associated with improved healthcare delivery. Arch Dis Child 85:386–390
Carcillo JA, Fields AI, American College of Critical Care Medicine Task Force Committee Members (2002) Clinical practice parameters for hemodynamic support of paediatric and neonatal patients in septic shock. Crit Care Med 30:1365–1378
Ceneviva G, Paschall JA, Maffei F, Carcillo JA (1998) Hemodynamic support in fluid-refractory paediatric septic shock. Paediatrics 102:1–6
Pizarro CF, Troster EJ, Damiani D, Carcillo JA (2005) Absolute and relative adrenal insufficiency in children with septic shock. Crit Care Med 33:855–859
Oliveira CF, Troster E, Oliveira DSF, Gottschald A, Moura J, Costa G, Vaz F, Carcillo JA, Rivers E (2007) An outcomes comparison of ACCM/PALS guidelines for paediatric septic shock with and without central venous oxygen saturation monitoring. Pediatr Crit Care Med 8:A237–A238
Carcillo JA, Hazelzet JA (2005) Sepsis and multiple organ system failure in children. In: Fink MP, Abraham E, Vincent JL, Kochanek PM (eds) Textbook of critical care. Elsevier Saunders, Philadelphia, pp 1267–1273
Oliveira CF, Troster EJ, Vaz FAC (2005) Description of technique for continuous monitoring of central venous oxygen saturation in infants and children with septic shock. Case reports. Revista Brasileira Terapia Intensiva 17:305–308
Wilkinson JD, Pollack MM, Ruttimann UE, Glass NL, Yeh TS (1986) Outcome of paediatric patients with multiple organ system failure. Crit Care Med 14:271–274
Wilkinson JD, Pollack MM, Glass NL, Kanter RK, Katz RW, Steinhart CM (1987) Mortality associated with multiple organ system failure and sepsis in paediatric intensive care unit. J Pediatr 111:324–328
Han YY, Carcillo JA, Dragotta MA, Bills DM, Watson RS, Westerman ME, Orr RA (2003) Early reversal of paediatric-neonatal septic shock by community physicians is associated with improved outcome. Paediatrics 112:793–799
Ninis N, Phillips C, Bailey L, Pollock JI, Nadel S, Britto J, Maconochie I, Winrow A, Coen PG, Booy R, Levin M (2005) The role of healthcare delivery in the outcome of meningococcal disease in children: case–control study of fatal and non-fatal cases. BMJ 330:1475
Rivers E, Nguyen B, Havstad S, Ressler J, Muzzin A, Knoblich B, Peterson E, Tomlanovich M, Early Goal-Directed Therapy Collaborative Group (2001) Early goal-directed therapy in the treatment of severe sepsis and septic shock. NEJM 345:1368–1377
Krafft P, Steltzer H, Hiesmayr M, Klimscha W, Hammerle AF (1993) Mixed venous oxygen saturation in critically ill septic shock patients. The role of defined events. Chest 103:900–906
Gattinoni L, Brazzi L, Pelosi P, Latini R, Tognoni G, Pesenti A, Fumagalli R (1995) A trial of goal-oriented hemodynamic therapy on critically ill patients. NEJM 333:1025–1032
Pearse RM, Rhodes A (2005) Mixed and central venous oxygen saturation. In: Vincent JL (ed) Yearbook of intensive care and emergency medicine. Springer, Berlin Heidelberg New York, pp 592–602
Scheinman MM, Brown MA, Rapaport E (1969) Critical assessment of use of central venous oxygen saturation as a mirror of mixed venous oxygen in severely ill cardiac patients. Circulation 40:165–172
Varpula M, Karlsson S, Ruokonen E, Pettila V (2006) Mixed venous oxygen saturation cannot be estimated by central venous oxygen saturation in septic shock. Intensive Care Med 32:1336–1343
Dueck MH, Klimek M, Appenrodt S, Weigand C, Boerner U (2005) Trends but not individual values of central venous oxygen saturation agree with mixed venous oxygen saturation during varying hemodynamic conditions. Anesthesiology 103:249–257
Chawla LS, Zia H, Gutierrez G, Katz NM, Seneff MG, Shah M (2004) Lack of equivalence between central and mixed venous oxygen saturation. Chest 126:1891–1896
Varpula M, Tallgren M, Saukkonen K, Voipio-Pulkki L-M, Pettilä V (2005) Hemodynamic variables related to outcome in septic shock. Intensive Care Med 31:1066–1071
Schranz D, Schmitt S, Oelert H, Schmid F, Huth R, Zimmer B, Schuind A, Vogel K, Stopfkuchen H, Jüngst BK (1989) Continuous monitoring of mixed venous oxygen saturation in infants after cardiac surgery. Intensive Care Med 15:228–232
Hirschl RB (1994) Oxygen delivery in the paediatric surgical patient. Curr Opin Pediatr 6:341–347
Estenssoro E, Gonzalez F, Laffaire E, Canales H, Sáenz G, Reina R, Dubin A (2005) Shock on admission day is the best predictor of prolonged mechanical ventilation in the ICU. Chest 127:598–603
Rossi C, Simini B, Brazzi L, Rossi G, Radrizzani D, Iapichino G, Bertolini G, Gruppo Italiano per la Valutazione degli Interventi in Terapia Intensiva (2006) Variable costs of ICU patients: a multicenter prospective study. Intensive Care Med 32:545–552
Odetola FO, Gebremariam A, Freed GL (2007) Patient and hospital correlates of clinical outcomes and resource utilization in severe pediatric sepsis. Pediatrics 119:487–494
Acknowledgements
We are indebted to the nursing and medical staff of the intensive care units; to Professor Claudio Leone, for statistical advice; and to Dr. Crésio Romeu Pereira, for assistance with study design.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is discussed in the editorial available at: http://dx.doi.org/10.1007/s00134-008-1086-8.
Trial registration: ClinicalTrials.gov identifier NCT00407823.
Conflicts of interest: In the past year, Dr. Rivers received research support from the National Institutes of Health, Hutchinson Technologies, Biosite Inc. and Edwards Lifesciences. Dr. Carcillo received funding from the National Institute of Health. The remaining authors have no conflict of interests to disclose.
Support: Supported by FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo – The State of São Paulo Research Foundation) and Edwards Lifesciences. FAPESP financed the acquisition of ScvO2 catheters, and Edwards Lifesciences provided ScvO2 monitors. Design and conduct of the study, collection, management, analysis and interpretation of the data, and preparation, review and approval of the manuscript, were done independently by the authors, who are not governed by the funding sponsors.
Rights and permissions
About this article
Cite this article
de Oliveira, C.F., de Oliveira, D.S.F., Gottschald, A.F.C. et al. ACCM/PALS haemodynamic support guidelines for paediatric septic shock: an outcomes comparison with and without monitoring central venous oxygen saturation. Intensive Care Med 34, 1065–1075 (2008). https://doi.org/10.1007/s00134-008-1085-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-008-1085-9